Durable, Complete Remission
For patients with mIDH1 AML who relapse or are refractory to initial treatment, choose REZLIDHIA as the first R/R targeted therapy1
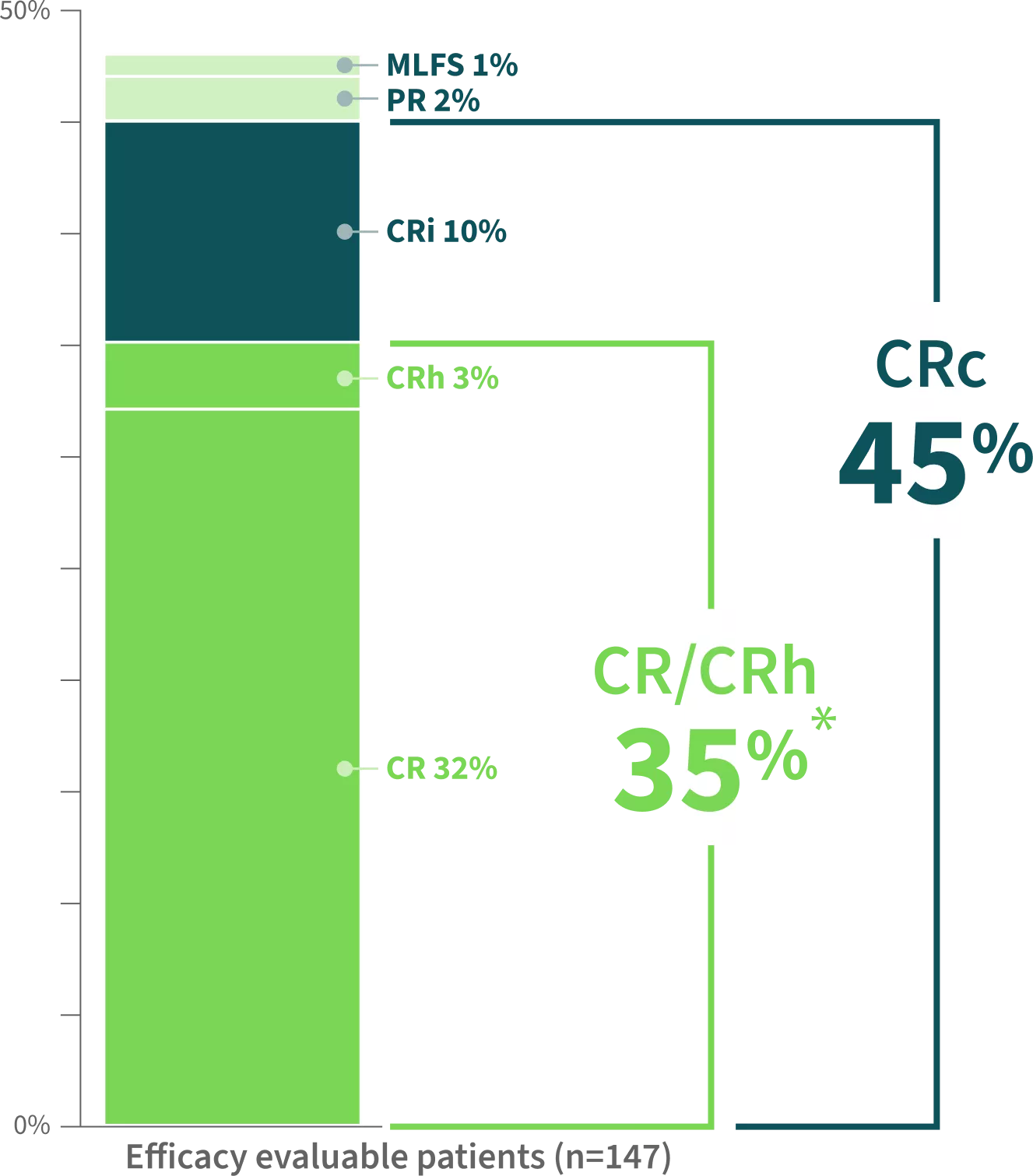
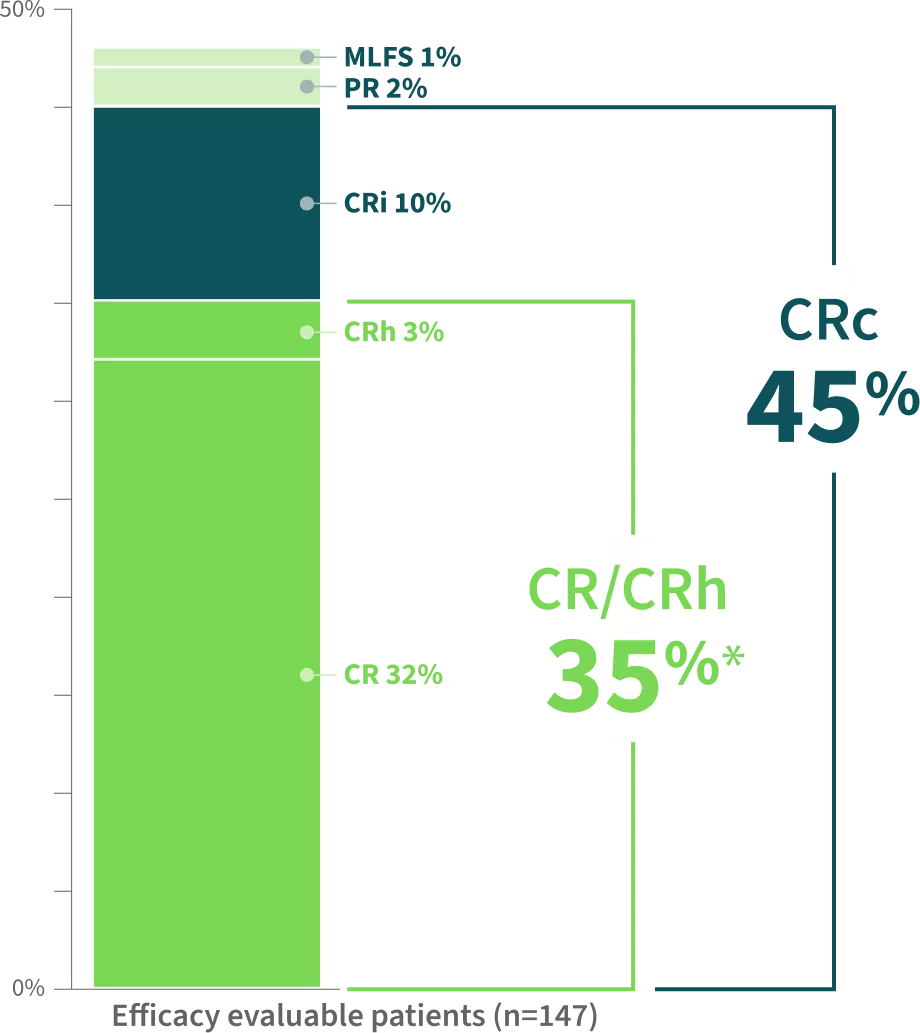
ALMOST HALF OF PATIENTS ACHIEVED A COMPOSITE COMPLETE REMISSION
Clinical Response Rates1-3
Additional Efficacy Outcomes1,2,4
- Response rates were similar regardless of prior treatment with venetoclax or intensive chemotherapy
- 11% (16 of 147) of patients underwent stem cell transplant after treatment with REZLIDHIA
The majority of responders achievedCOMPLETE REMISSION2
Data cutoff=June 15, 2023.
Primary endpoint.1
AML=acute myeloid leukemia; CR=complete remission; CRc=composite complete remission (CR+CRh+CRi); CRh=complete remission with partial hematologic recovery; CRi=complete remission with incomplete blood count recovery; mIDH1=mutated isocitrate dehydrogenase-1; MLFS=morphologic leukemia–free state; PR=partial remission (which required recovery of both neutrophil and platelet counts consistent with a CR); R/R=relapsed/refractory.
MEDIAN DURATION OF RESPONSE IN PATIENTS ACHIEVING CR/CRh WAS GREATER THAN 2 YEARS
Duration of CR/CRh Response3
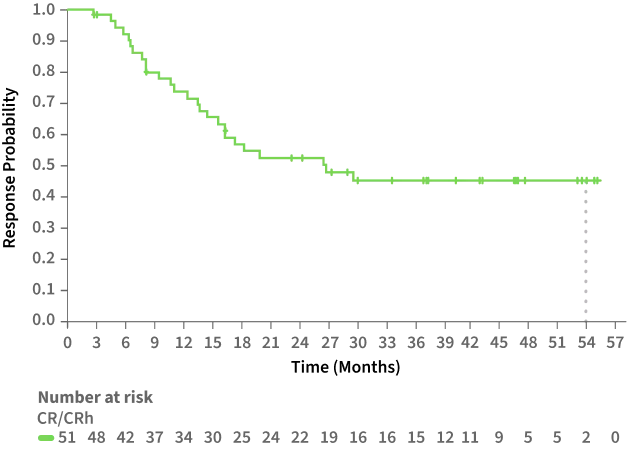
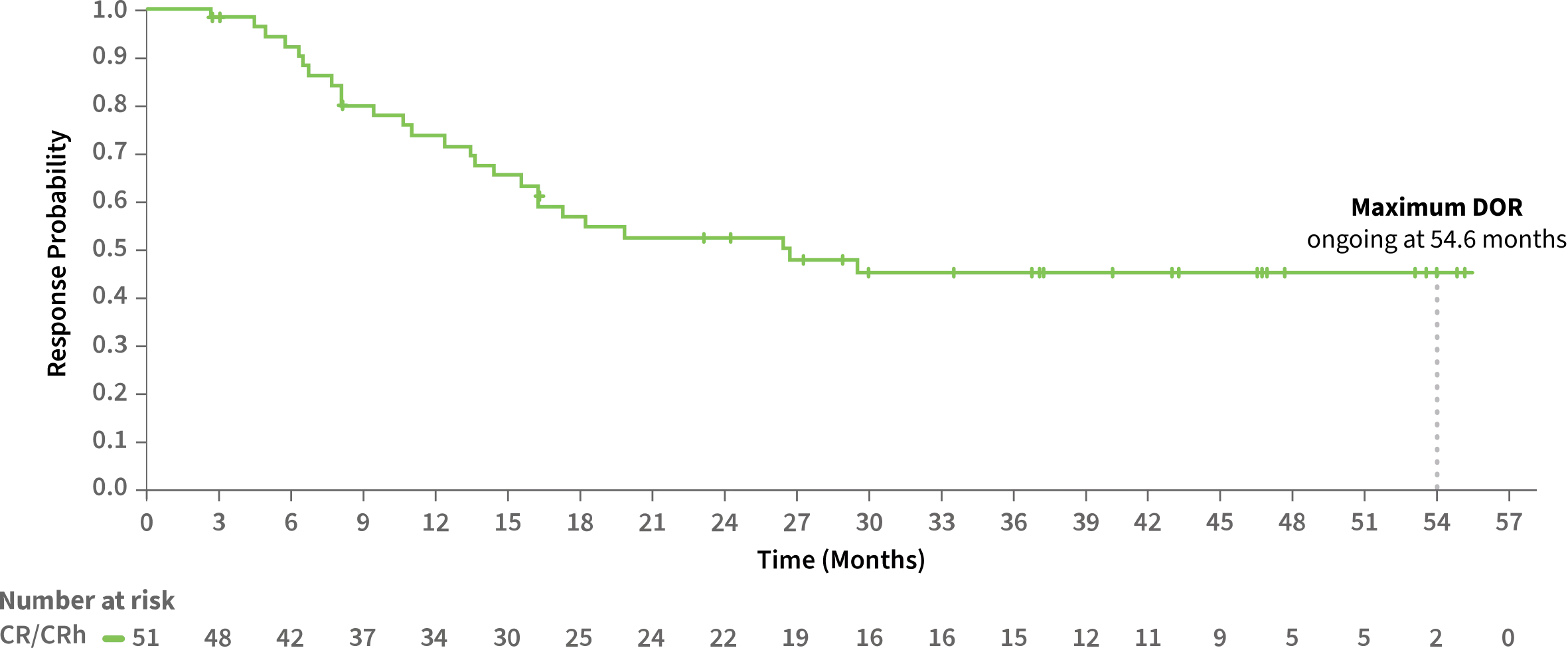
Median duration of CR/CRh:
25.3 MONTHS
(95% CI: 13.5-NR)
CI=confidence interval; DOR=duration of response; NR=not reached.
REZLIDHIA RESPONSE RATES AS THE FIRST IDH1 INHIBITOR FOLLOWING STANDARD OF CARE TREATMENT PROGRESSION
Subgroup analysis after a venetoclax-based regimen or intensive chemotherapy
Prior Venetoclax-based Regimen (n=12)5
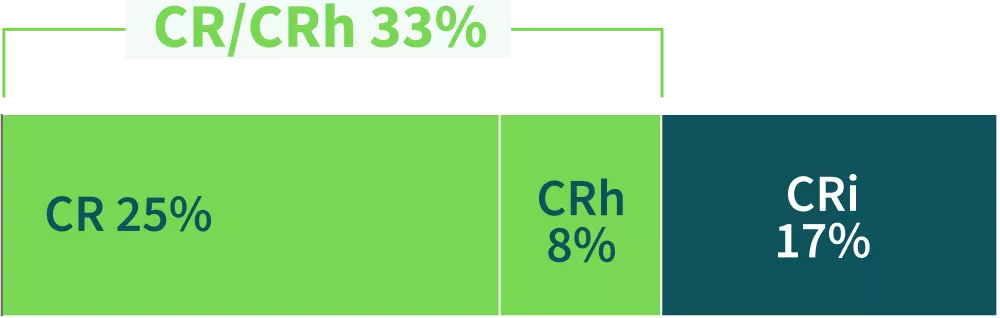
Median duration of previous venetoclax-based treatment was 6.7 months (range: 2-34 months)6

CRC=50%5
Median duration of CR/CRh4: NOT REACHED (95% CI: 4.8 months-NR)
Median duration of previous venetoclax-based treatment was 6.7 months (range: 2-34 months)6
Prior Intensive Chemotherapy (n=13)4*
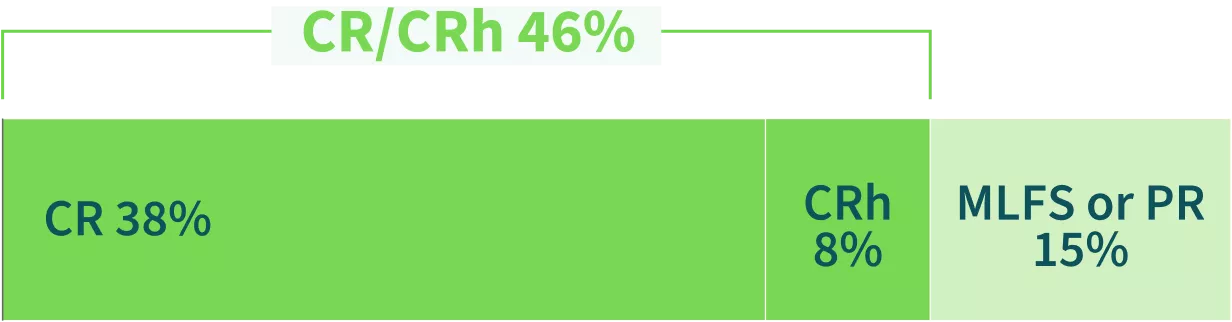

CRC=46%4
Median duration of CR/CRh4: NOT REACHED (95% CI: 1.8 months-NR)
Intensive chemotherapy was the only prior regimen these patients received. Intensive chemotherapy was defined as high-dose cytarabine with daunorubicin, mitoxantrone with etoposide and cytarabine, or transplant conditioning regimens, whether myeloablative or not.
IDH1=isocitrate dehydrogenase-1.
Results in these high-risk subgroups are consistent
with the overall patient population (N=147):
45% achieved CRc2-5,7,8
MEDIAN TIME TO CR/CRh WAS APPROXIMATELY 2 MONTHS WITH REZLIDHIA
Time to Response in Patients Achieving CR/CRh (n=51)3
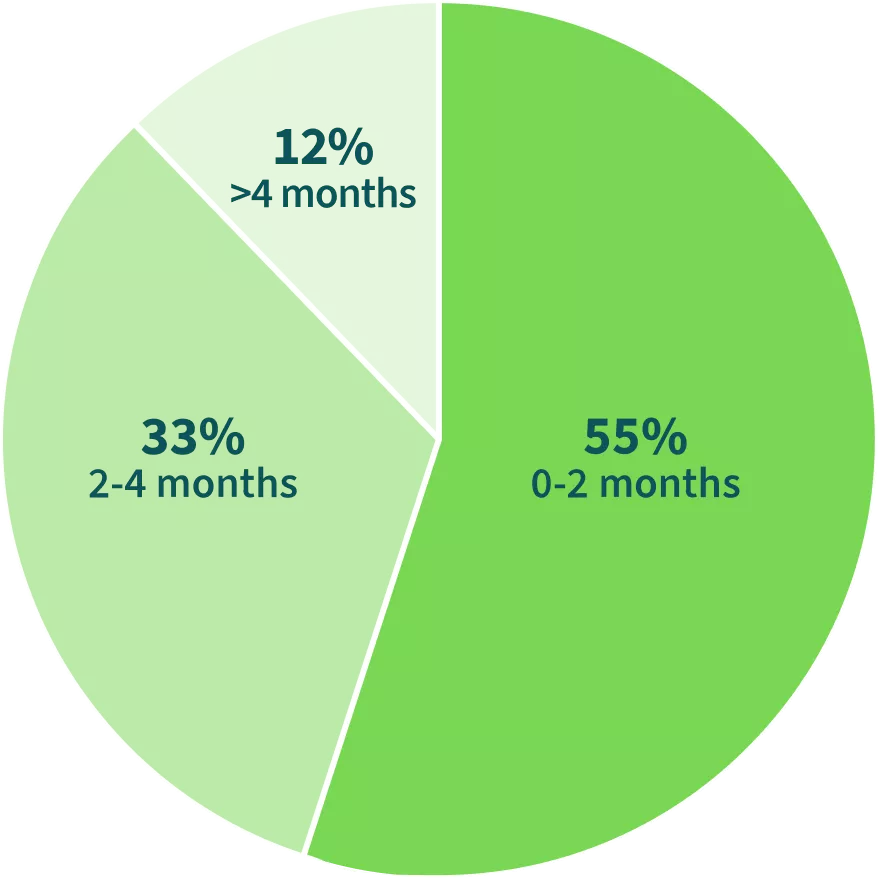
88% RESPONDED
within 4 months
Median time to CR/CRh:
1.9 MONTHS
(95% CI: 1-2.8)3,4
For patients without disease progression or unacceptable toxicity, treat for a minimum of 6 months to allow time for clinical response1
Results in patients who received prior venetoclax were also promising; the remission rate was consistent with that of the broader study population, and most patients who achieved remission had an ongoing remission at the time of data cutoff.
—de Botton et al. Blood Adv. 2023.2
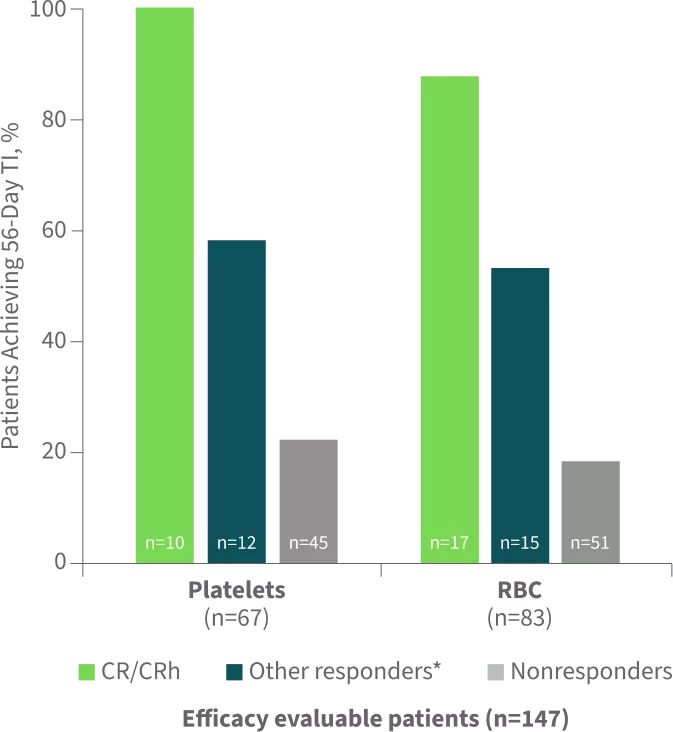
IMPROVEMENT IN TRANSFUSION INDEPENDENCE WAS SEEN ACROSS ALL PATIENT GROUPS
34% of transfusion-dependent patients became transfusion independent1
Patient Outcomes1
- Of the 86 patients who were transfusion dependent at baseline, a 56-day TI was achieved in 29 (34%), including patients in all response groups
- Of the 61 patients who were independent of both RBC and platelet transfusions at baseline, 39 (64%) remained transfusion independent
Percentage of Patients Achieving ≥56-Day Transfusion Independence (TI)2
Percentage of Patients Achieving ≥56-Day Transfusion Independence (TI)2
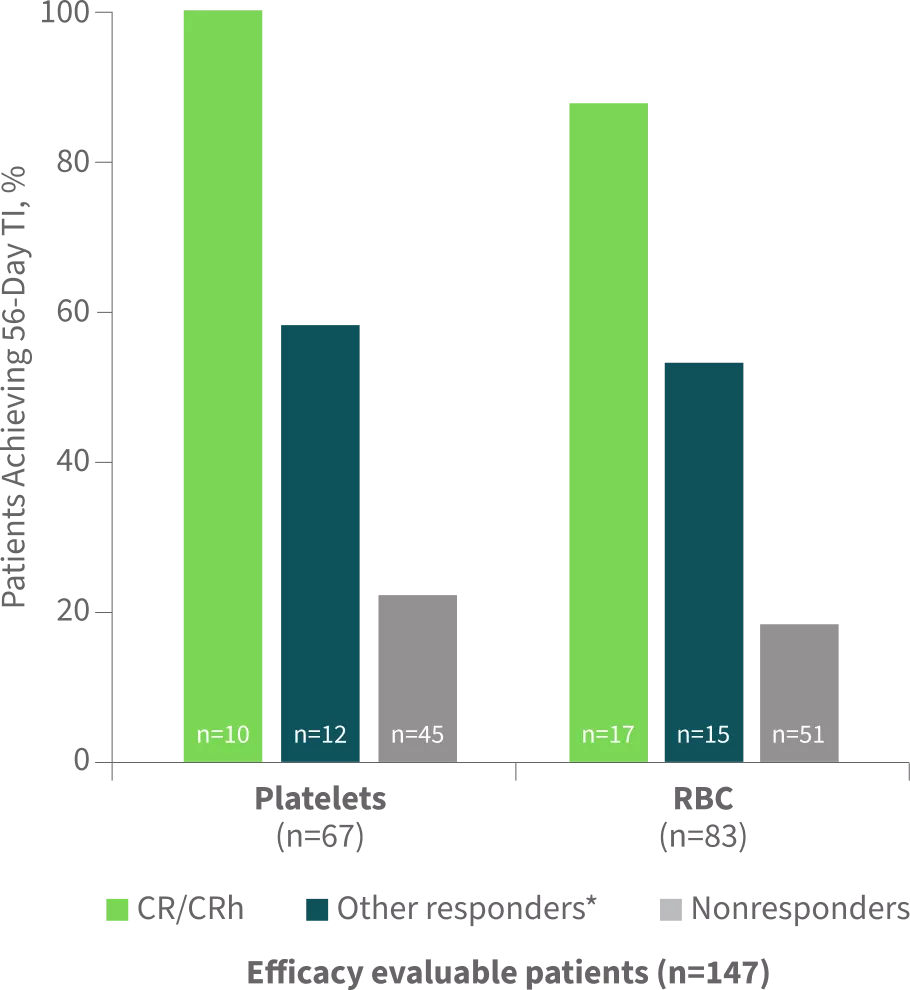
Data cutoff=June 18, 2021.
Other responders include patients achieving CRi, PR, or MLFS.2
Data cutoff=June 18, 2021.
Other responders include patients achieving CRi, PR, or MLFS.2
The observed efficacy is clinically meaningful and represents a therapeutic advance in this molecularly defined patient population with a poor prognosis and limited treatment options.
—de Botton et al. Blood Adv. 2023.2
CLINICAL TRIAL: STUDY 2102‑HEM‑101
REZLIDHIA was studied in an open-label, single-arm, multicenter trial of patients representative of those seen in clinical practice.1,2
Patients were enrolled between April 2018 and June 2020. Initial data cutoff=June 18, 2021. Final data cutoff=June 15, 2023.
| Patient Population (N=153) |
|---|
Adults (≥18 years old) Relapsed/refractory AML Confirmed IDH1 mutation No prior IDH1-inhibitor therapy ECOG PS 0-2 |
| Dosing |
REZLIDHIA* 150 mg Twice daily, orally Treated until disease progression |
| Clinical Endpoints |
Primary Endpoint Rate of CR/CRh† Secondary Endpoints
|
| Patient Population (N=153) | Dosing | Clinical Endpoints |
|---|---|---|
Adults (≥18 years old) Relapsed/refractory AML Confirmed IDH1 mutation No prior IDH1-inhibitor therapy ECOG PS 0-2 | REZLIDHIA* 150 mg Twice daily, orally Treated until disease progression | Primary Endpoint Rate of CR/CRh† Secondary Endpoints
|
REZLIDHIA was taken by mouth twice daily over continuous 28-day cycles, with doses at least 8 hours apart on an empty stomach.
Defined as bone marrow blasts <5% with absolute neutrophil count >500/microliter and platelet count >50,000/microliter.
DOR was calculated from the time of first response until death, relapse, or new anti-cancer therapy; patients without an event were censored at their last response assessment. Patients who discontinued therapy to proceed to hematopoietic stem cell transplantation (HSCT) were followed for DOR and OS, as HSCT was not an event.
Patients were classified as “transfusion dependent” if platelet and/or red blood cell (RBC) transfusion occurred within 56 days prior to baseline, and were transfusion independent if without platelet and/or RBC transfusions for at least 56 days during treatment.
ECOG PS=Eastern Cooperative Oncology Group Performance Status.
| Patient Demographic and Baseline Disease Characteristics | Efficacy-Evaluable Population (N=147) |
|---|
Demographics Age (Years), Median (Min, Max) 71 (32, 87) Age Categories, n (%) <65 years 37 (25) ≥65 years to <75 years 65 (44) ≥75 years 45 (31) Sex, n (%) Male 74 (50) Female 73 (50) Disease Characteristics ECOG PS, n (%) 0 45 (31) 1 76 (52) 2 23 (16) IDH1 Mutation, n (%)* R132C 85 (58) R132H 35 (24) R132G 12 (8) R132S 11 (7) R132L 4 (3) Comutations, n (%)† NPM1 31 (21) FLT3 15 (10) TP53 9 (6) Type of AML, n (%) De novo AML 97 (66) Secondary AML 50 (34) Cytogenetic Risk Status, n (%)‡ Favorable 6 (4) Intermediate 107 (73) Poor 25 (17) Unknown 9 (6) | Disease Characteristics (Cont’d) Prior AML Therapy Outcome, n (%) Refractory 51 (35) Relapsed 96 (65) Remission duration 67 (70) Remission duration 29 (30) Relapse Patients, n (%) 1 87 (59) 2 11 (8) ≥3 3 (2)
Transfusion Dependent at 86 (59) Prior Treatments Number of Prior Treatments Median (range) 2 (1-7) 1 regimen, n (%) 48 (33) 2 regimens, n (%) 45 (31) ≥3 regimens, n (%) 54 (37) Prior Treatments Received, n (%)‖ Cytarabine 105 (71) Idarubicin 64 (44) Daunorubicin 31 (21) Fludarabine 25 (17) Hypomethylating agent 58 (39) As a single agent 21 (14) Gemtuzumab-based 11 (7) Venetoclax-based 12 (8) With a hypomethylating 8 (5)
Prior Stem Cell Transplantation 17 (12) |
Using central IDH1 assay testing results.
Mutations other than IDH1.
Cytogenetic risk categorization was investigator reported by National Comprehensive Cancer Network® (NCCN®) or European LeukemiaNet (ELN) guidelines.
Transfusion dependent at baseline is defined as receiving a transfusion within 56 days prior to first dose of olutasidenib or noting transfusion dependence prior to coming on study.
FLT3=FMS-like tyrosine kinase 3; NPM1=nucleophosmin 1; TP53=tumor protein p53.

Olutasidenib (REZLIDHIA®) is recommended by the National Comprehensive Cancer Network® (NCCN®) as a targeted treatment option for relapsed/refractory AML with an IDH1 mutation.9
References:
- REZLIDHIA®. Package insert. Rigel Pharmaceuticals, Inc; 2022.
- de Botton S, Fenaux P, Yee K, et al. Olutasidenib (FT-2102) induces durable complete remissions in patients with relapsed or refractory IDH1-mutated AML. Blood Adv. 2023;7(13):3117-3127. doi:10.1182/bloodadvances.2022009411
- Cortes J, Curti A, Fenaux P, et al. Olutasidenib for mutated IDH1 acute myeloid leukemia: final five-year results from the phase 2 pivotal cohort. J Hematol Oncol. 2025;18(1):102. doi:10.1186/s13045-025-01751-w
- Data on file, Rigel Pharmaceuticals, Inc. January 2025.
- Cortes J, Curti A, Fenaux P, et al. Olutasidenib for mutated IDH1 acute myeloid leukemia: final five-year results from the phase 2 pivotal cohort (supplemental data). J Hematol Oncol. 2025; https://link.springer.com/article/10.1186/s13045-025-01751-w#Sec20
- Data on file, Rigel Pharmaceuticals, Inc. January 2024.
- Maiti A, Rausch CR, Cortes JE, et al. Outcomes of relapsed or refractory acute myeloid leukemia after frontline hypomethylating agent and venetoclax regimens. Haematologica. 2021;106(3):894-898. doi:10.3324/haematol.2020.252569
- Niscola P, Gianfelici V, Catalano G, et al. Acute Myeloid Leukemia in Older Patients: From New Biological Insights to Targeted Therapies. Curr Oncol. 2024;31:6632–6658. https://doi.org/10.3390/curroncol31110490
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Acute Myeloid Leukemia V.3.2026. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed December 11, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
INDICATION AND IMPORTANT SAFETY INFORMATION, INCLUDING BOXED WARNING
REZLIDHIA is indicated for the treatment of adult patients with relapsed or refractory acute myeloid leukemia (AML) with a susceptible isocitrate dehydrogenase-1 (IDH1) mutation as detected by an FDA-approved test.
WARNING: DIFFERENTIATION SYNDROME Differentiation syndrome, which can be fatal, can occur with REZLIDHIA treatment. Symptoms may include dyspnea, pulmonary infiltrates/pleuropericardial effusion, kidney injury, hypotension, fever, and weight gain. If differentiation syndrome is suspected, withhold REZLIDHIA and initiate treatment with corticosteroids and hemodynamic monitoring until symptom resolution.
WARNINGS AND PRECAUTIONS
Differentiation Syndrome REZLIDHIA can cause differentiation syndrome. In the clinical trial of REZLIDHIA in patients with relapsed or refractory AML, differentiation syndrome occurred in 16% of patients, with grade 3 or 4 differentiation syndrome occurring in 8% of patients treated, and fatalities in 1% of patients. Differentiation syndrome is associated with rapid proliferation and differentiation of myeloid cells and may be life-threatening or fatal. Symptoms of differentiation syndrome in patients treated with REZLIDHIA included leukocytosis, dyspnea, pulmonary infiltrates/pleuropericardial effusion, kidney injury, fever, edema, pyrexia, and weight gain. Of the 25 patients who experienced differentiation syndrome, 19 (76%) recovered after treatment or after dose interruption of REZLIDHIA. Differentiation syndrome occurred as early as 1 day and up to 18 months after REZLIDHIA initiation and has been observed with or without concomitant leukocytosis.
If differentiation syndrome is suspected, temporarily withhold REZLIDHIA and initiate systemic corticosteroids (e.g., dexamethasone 10 mg IV every 12 hours) for a minimum of 3 days and until resolution of signs and symptoms. If concomitant leukocytosis is observed, initiate treatment with hydroxyurea, as clinically indicated. Taper corticosteroids and hydroxyurea after resolution of symptoms. Differentiation syndrome may recur with premature discontinuation of corticosteroids and/or hydroxyurea treatment. Institute supportive measures and hemodynamic monitoring until improvement; withhold dose of REZLIDHIA and consider dose reduction based on recurrence.
Hepatotoxicity REZLIDHIA can cause hepatotoxicity, presenting as increased alanine aminotransferase (ALT), increased aspartate aminotransferase (AST), increased blood alkaline phosphatase, and/or elevated bilirubin. Of 153 patients with relapsed or refractory AML who received REZLIDHIA, hepatotoxicity occurred in 23% of patients; 13% experienced grade 3 or 4 hepatotoxicity. One patient treated with REZLIDHIA in combination with azacitidine in the clinical trial, a combination for which REZLIDHIA is not indicated, died from complications of drug-induced liver injury. The median time to onset of hepatotoxicity in patients with relapsed or refractory AML treated with REZLIDHIA was 1.2 months (range: 1 day to 17.5 months) after REZLIDHIA initiation, and the median time to resolution was 12 days (range: 1 day to 17 months). The most common hepatotoxicities were elevations of ALT, AST, blood alkaline phosphatase, and blood bilirubin.
Monitor patients frequently for clinical symptoms of hepatic dysfunction such as fatigue, anorexia, right upper abdominal discomfort, dark urine, or jaundice. Obtain baseline liver function tests prior to initiation of REZLIDHIA, at least once weekly for the first two months, once every other week for the third month, once in the fourth month, and once every other month for the duration of therapy. If hepatic dysfunction occurs, withhold, reduce, or permanently discontinue REZLIDHIA based on recurrence/severity.
ADVERSE REACTIONS
The most common (≥20%) adverse reactions, including laboratory abnormalities, were aspartate aminotransferase increased, alanine aminotransferase increased, potassium decreased, sodium decreased, alkaline phosphatase increased, nausea, creatinine increased, fatigue/malaise, arthralgia, constipation, lymphocytes increased, bilirubin increased, leukocytosis, uric acid increased, dyspnea, pyrexia , rash, lipase increased, mucositis, diarrhea and transaminitis.
DRUG INTERACTIONS
- Avoid concomitant use of REZLIDHIA with strong or moderate CYP3A inducers.
- Avoid concomitant use of REZLIDHIA with sensitive CYP3A substrates unless otherwise instructed in the substrates prescribing information. If concomitant use is unavoidable, monitor patients for loss of therapeutic effect of these drugs.
LACTATION
Advise women not to breastfeed during treatment with REZLIDHIA and for 2 weeks after the last dose.
GERIATRIC USE
No overall differences in effectiveness were observed between patients 65 years and older and younger patients. Compared to patients younger than 65 years of age, an increase in incidence of hepatotoxicity and hypertension was observed in patients ≥65 years of age.
HEPATIC IMPAIRMENT
In patients with mild or moderate hepatic impairment, closely monitor for increased probability of differentiation syndrome.
Please see Full Prescribing Information, including Boxed WARNING.
REZLIDHIA is indicated for the treatment of adult patients with relapsed or refractory acute myeloid leukemia (AML) with a susceptible isocitrate dehydrogenase-1 (IDH1) mutation as detected by an FDA-approved test.
WARNING: DIFFERENTIATION SYNDROME Differentiation syndrome, which can be fatal, can occur with REZLIDHIA treatment. Symptoms may include dyspnea, pulmonary infiltrates/pleuropericardial effusion, kidney injury, hypotension, fever, and weight gain. If differentiation syndrome is suspected, withhold REZLIDHIA and initiate treatment with corticosteroids and hemodynamic monitoring until symptom resolution.
WARNINGS AND PRECAUTIONS
Differentiation Syndrome REZLIDHIA can cause differentiation syndrome. In the clinical trial of REZLIDHIA in patients with relapsed or refractory AML, differentiation syndrome occurred in 16% of patients, with grade 3 or 4 differentiation syndrome occurring in 8% of patients treated, and fatalities in 1% of patients. Differentiation syndrome is associated with rapid proliferation and differentiation of myeloid cells and may be life-threatening or fatal. Symptoms of differentiation syndrome in patients treated with REZLIDHIA included leukocytosis, dyspnea, pulmonary infiltrates/pleuropericardial effusion, kidney injury, fever, edema, pyrexia, and weight gain. Of the 25 patients who experienced differentiation syndrome, 19 (76%) recovered after treatment or after dose interruption of REZLIDHIA. Differentiation syndrome occurred as early as 1 day and up to 18 months after REZLIDHIA initiation and has been observed with or without concomitant leukocytosis.
If differentiation syndrome is suspected, temporarily withhold REZLIDHIA and initiate systemic corticosteroids (e.g., dexamethasone 10 mg IV every 12 hours) for a minimum of 3 days and until resolution of signs and symptoms. If concomitant leukocytosis is observed, initiate treatment with hydroxyurea, as clinically indicated. Taper corticosteroids and hydroxyurea after resolution of symptoms. Differentiation syndrome may recur with premature discontinuation of corticosteroids and/or hydroxyurea treatment. Institute supportive measures and hemodynamic monitoring until improvement; withhold dose of REZLIDHIA and consider dose reduction based on recurrence.
Hepatotoxicity REZLIDHIA can cause hepatotoxicity, presenting as increased alanine aminotransferase (ALT), increased aspartate aminotransferase (AST), increased blood alkaline phosphatase, and/or elevated bilirubin. Of 153 patients with relapsed or refractory AML who received REZLIDHIA, hepatotoxicity occurred in 23% of patients; 13% experienced grade 3 or 4 hepatotoxicity. One patient treated with REZLIDHIA in combination with azacitidine in the clinical trial, a combination for which REZLIDHIA is not indicated, died from complications of drug-induced liver injury. The median time to onset of hepatotoxicity in patients with relapsed or refractory AML treated with REZLIDHIA was 1.2 months (range: 1 day to 17.5 months) after REZLIDHIA initiation, and the median time to resolution was 12 days (range: 1 day to 17 months). The most common hepatotoxicities were elevations of ALT, AST, blood alkaline phosphatase, and blood bilirubin.
Monitor patients frequently for clinical symptoms of hepatic dysfunction such as fatigue, anorexia, right upper abdominal discomfort, dark urine, or jaundice. Obtain baseline liver function tests prior to initiation of REZLIDHIA, at least once weekly for the first two months, once every other week for the third month, once in the fourth month, and once every other month for the duration of therapy. If hepatic dysfunction occurs, withhold, reduce, or permanently discontinue REZLIDHIA based on recurrence/severity.
ADVERSE REACTIONS
The most common (≥20%) adverse reactions, including laboratory abnormalities, were aspartate aminotransferase increased, alanine aminotransferase increased, potassium decreased, sodium decreased, alkaline phosphatase increased, nausea, creatinine increased, fatigue/malaise, arthralgia, constipation, lymphocytes increased, bilirubin increased, leukocytosis, uric acid increased, dyspnea, pyrexia, rash, lipase increased, mucositis, diarrhea and transaminitis.
DRUG INTERACTIONS
- Avoid concomitant use of REZLIDHIA with strong or moderate CYP3A inducers.
- Avoid concomitant use of REZLIDHIA with sensitive CYP3A substrates unless otherwise instructed in the substrates prescribing information. If concomitant use is unavoidable, monitor patients for loss of therapeutic effect of these drugs.
LACTATION
Advise women not to breastfeed during treatment with REZLIDHIA and for 2 weeks after the last dose.
GERIATRIC USE
No overall differences in effectiveness were observed between patients 65 years and older and younger patients. Compared to patients younger than 65 years of age, an increase in incidence of hepatotoxicity and hypertension was observed in patients ≥65 years of age.
HEPATIC IMPAIRMENT
In patients with mild or moderate hepatic impairment, closely monitor for increased probability of differentiation syndrome.
Please see Full Prescribing Information, including Boxed WARNING.